The enthusiasm for potential therapeutic use of GLP-1 derives from studies demonstrating that unlike GIP, the glucose-lowering actions of GLP-1 are preserved in patients with type 2 diabetes, as shown in Preserved incretin activity of glucagon-like peptide 1 [7-36 amide] but not of synthetic human gastric inhibitory polypeptide in patients with type-2 diabetes mellitus. J Clin Invest. 1993 Jan;91(1):301-7 and The insulinotropic actions of glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (7-37) in normal and diabetic subjects.
Similarly, the actions of GLP-1 on inhibition of gastric emptying are also preserved in subjects with poorly controlled type 2 diabetes, Gastric emptying, glucose responses, and insulin secretion after a liquid test meal: effects of exogenous glucagon-like peptide-1 (GLP-1)-(7-36) amide in type 2 (noninsulin-dependent) diabetic patients. J Clin Endocrinol Metab. 1996 Jan;81(1):327-32.
Although the actions of GLP-1 on the b-cell are preserved yet modestly diminished in T2DM, the diabetic a-cell retains near normal responsivity to low dose GLP-1 infusion, with inhibition of glucagon secretion seen to a similar extent in diabetic vs. non-diabetic subjects. Hence, modestly diminished GLP-1 action in diabetic subjects does not likely contribute to the defective glucose-stimulated glucagon suppression that remains a characteristic of diabetic subjects. See Preserved inhibitory potency of GLP-1 on glucagon secretion in type 2 diabetes mellitus. J Clin Endocrinol Metab. 2009 Dec;94(12):4679-87. Epub 2009 Oct 16
Hart and colleagues used the Metabochip to identify genetic variants affecting GLP-1 induced insulin secretion during hyperglycemic clamps in 232 nondiabetic participants from two independent populations, the Netherlands Twin Register (NTR) and the German Tübingen cohort. The predictive value of these SNPs was then assessed in a second population of 527 subjects with T2DM treated with either GLP-1R agonists or DPP-4 inhibitors for at least 3 months. Three distinct genetic loci were identified that associated with a reduced magnitude of insulin secretion in response to GLP-1 in non-diabetic subjects; rs4148941 is located in the 3’-untranslated region of the second exon of the CHST3 (Carbohydrate (Chondroitin 6) Sulfotransferase 3 gene, with carriers of the C allele exhibing a 32% reduction in GLP-1-stimulated insulin secretion. Homozygous carriers of Homozygous carriers of the minor allele of rs7202633, 84kb upstream of TMEM114, exhibited a 2-fold increase in GLP-1-stimulated insulin secretion. The third (G) allele, rs7202877 near CTRB1, CTRB2 and BCAR1, was associated with a 33% increase in GLP-1-stimulated insulin secretion. None of these alleles exhibited alterations in glucose-stimulated insulin secretion. Diabetic patients carrying the G allele of rs7202877 near CTRB1/2 showed a significantly smaller (0.51%) decrease in A1C levels after DPP-4 inhibitor treatment compared to TT carriers. In contrast, these same alleles did not predict the response to therapy with GLP-1R agonists. The rs7202877 G allele was associated with an increased chymotrypsin activity in stool samples and increased islet expression of CTRB1 and CTRB2. The mechanisms linking chymotrypsin biology to altered incretin responsibity require further clarification. The CTRB1/2 locus affects diabetes susceptibility and treatment via the incretin pathway Diabetes. 2013 May 14. Diabetes. 2013 May 14.
Jones and colleagues identified features associated with better or worse A1c reductions in response to therapy with GLP-1R agonists. Duration of diabetes, reduced baseline parameters of beta cell function and positive GAD or IA2 autoantibodies, together with relative insulin deficiency, predicted a suboptimal therapeutic response to GLP-1R agonist therapy assessed over 6 months Markers of β-Cell Failure Predict Poor Glycemic Response to GLP-1 Receptor Agonist Therapy in Type 2 Diabetes Diabetes Care. 2015 Aug 4. pii: dc150258.
Whether endogenous GLP-1R signaling is similarly important for beta-cell function has been examined using exendin(9-39) to block endogenous GLP-1 action in subjects with type 2 diabetes. The increase in postprandial insulin secretion was attenuated by exendin(9-39) in both normal subjects and in patients with T2DM. Intriguingly, exendin(9-39) also reduced the insulin response to iv glucose challenge, under circumstances where circulating levels of GLP-1 were so low so as to be undetectable. Moreover, glucagon levels were increased during the latter component of the meal following exendin (9-39) infusion in diabetic subjects. These findings indicate that even very low basal levels of GLP-1R signaling are important for b-cell and islet function in response to glucose in humans, perhaps under some circumstances through neural pathways. Moreover, the endogenous GLP-1 axis does not seem to be compromised in this small set of diabetic subjects. See Effect of endogenous GLP-1 on insulin secretion in type 2 diabetes. Diabetes. 2010 Mar 9. [Epub ahead of print].
Although the total number of diabetic patients treated with native GLP-1 to date remains small, combined analyses of several studies shows uniform responses to GLP-1 with glucose lowering in all diabetic subjects analyzed to date. See Glucagon-like peptide 1 and its potential in the treatment of non-insulin-dependent diabetes mellitus. Horm Metab Res. 1997 Sep;29(9):411-6. Nevertheless, it seems clear that there is a spectrum of GLP-1 responses in subjects with Type 2 diabetes, with the greatest 'GLP-1 effectiveness' observed in short term intravenous infusion studies, in subjects with more modest impairment in fasting hyperglycemia. See Determinants of the effectiveness of glucagon-like peptide-1 in type 2 diabetes. J Clin Endocrinol Metab. 2001 Aug;86(8):3853-60
Similarly, although long term data for use of native GLP-1 in the treatment of diabetics is currently lacking, treatment of diabetic patients for 3-6 weeks with subcutaneous GLP-1 was efficacious in several small pilot studies, Subcutaneous glucagon-like peptide-1 improves postprandial glycaemic control over a 3-week period in patients with early type 2 diabetes. Clin Sci (Colch). 1998 Sep;95(3):325-9 and Glucagon-like peptide-1 (GLP-1): a trial of treatment in non-insulin -dependent diabetes mellitus. Eur J Clin Invest. 1997 Jun;27(6):533-6.
For results of longer term studies in human subjects, see, GLP-1 analogues and human diabetes
To review safety issues surrounding use of GLP-1R agonists to treat diabetes, see GLP-1: Adverse Events
Importantly, GLP-1 lowers blood glucose in the overnight fasting state, and not just after meal ingestion, as shown in Normalization of fasting hyperglycaemia by exogenous glucagon-like peptide 1 (7-36 amide) in type 2 (non-insulin-dependent) diabetic patients. Diabetologia. 1993 Aug;36(8):741-4 and Normalization of insulin responses to glucose by overnight infusion of glucagon-like peptide 1 (7-36) amide in patients with NIDDM. Diabetes. 1996 Nov;45(11):1524-30, largely via improvement of β cell function. It seems clear that although the predominant gluose-lowering effects of GLP-1R agonists are during the day, studies examining 16 hr vs 24 hr infusion of GLP-1 in hospitalized patients for 7 days clearly demonstrated better 24 hr glucose control in subjects receiving the continuous 24 hr infusion as shown in Glucagon-like peptide-1 infusion must be maintained for 24 h/day to obtain acceptable glycemia in type 2 diabetic patients who are poorly controlled on sulphonylurea treatment. Diabetes Care. 2001 Aug;24(8):1416-21
The finding that short-term GLP-1 infusion normalized fasting plasma glucose in patients with type 2 diabetes following secondary sulfonylurea failure suggests that activation of GLP-1R signaling may reverse diabetes-associated defects in the failing β cell, as shown in Influence of glucagon-like peptide 1 on fasting glycemia in type 2 diabetic patients treated with insulin after sulfonylurea secondary failure. Diabetes Care. 1998 Nov;21(11):1925-31. Furthermore, the glucose-lowering effects of GLP-1 and sulfonylureas are additive, as shown in Glucagon-like peptide I enhances the insulinotropic effect of glibenclamide in NIDDM patients and in the perfused rat pancreas. Diabetes Care. 1996 Aug;19(8):857-63.
Infusion of GLP-1 for varying periods of time produces different results on β cell function, consistent with the notion that GLP-1R activation sets in motion a large number of molecular events in addition to acute stimulation of exocytosis and insulin secretion. A three hour GLP-1 infusion produced more significant improvement in first phase insulin secretion and second phase insulin release compared to a single acute GLP-1 injection. See Differential Effects of Acute and Extended Infusions of Glucagon-Like Peptide-1 on First- and Second-Phase Insulin Secretion in Diabetic and Nondiabetic Humans. Diabetes Care. 2003 Mar;26(3):791-798
Although numerous studies attest to the effectiveness of GLP-1 in the acute lowering of blood glucose in subjects with type 2 diabetes, graded infusion of varying glucose and GLP-1 concentrations in seven diabetic subjects demonstrates a clearly detectable difference in β cell responsivity to GLP-1 in diabetic subjects. β cell responsivity, as measured by the insulin secretory response, was clearly lower in subjects with Type 2 diabetes compared to normal controls, but improves significantly following even low dose GLP-1 infusion. See The Influence of GLP-1 on Glucose-Stimulated Insulin Secretion: Effects on beta-Cell Sensitivity in Type 2 and Nondiabetic Subjects. Diabetes. 2003 Feb;52(2):380-386. Nevertheless, GLP-1 infusion potentiates the acute β cell secretory response to glucose after meal ingestion in human subjects as shown in Characterization of GLP-1 Effects on beta-Cell Function After Meal Ingestion in Humans. Diabetes Care. 2003 Oct;26(10):2860-4.
Does GLP-1 work additively in the presence of other antidiabetic agents? The answer, at least for metformin, appears to be yes. See Additive glucose-lowering effects of glucagon-like peptide-1 and metformin in type 2 diabetes. Diabetes Care. 2001 Apr;24(4):720-5
Similarly, addition of 48 hrs of GLP-1 therapy in diabetic subjects previously treated with the thiazolidinedione pioglitazone increased levels of insulin, decreased plasma glucagon and produced further reductions in fasting plasma glucose, 8 hr mean plasma glucose, and levels of free fatty acids. See Additive effects of glucagon-like Peptide 1 and pioglitazone in patients with type 2 diabetes. Diabetes Care. 2004 Aug;27(8):1910-4.
Can GLP-1 be delivered via a non-injectable formulation? Several studies appear promising, however additional refinements in these technologies are eagerly awaited. See GLP-1 tablet in type 2 diabetes in fasting and postprandial conditions. Diabetes Care. 1997 Dec;20(12):1874-9
Does it matter greatly whether GLP-1 is administered prior to or after food intake for the treatment of Type 2 diabetes? At least in the very short term single injection paradigm, the answer is no. Although the mechanism for antidiabetogenic action of GLP-1 may be different (gastric emptying versus effects on glucagon and insulin), some degree of postprandial glycemic control was achieved with various pre and postmeal GLP-1 administration regimens. More long term studies (24h to several days and weeks) are required before these conclusions can be more widely extrapolated. See Antidiabetogenic action of glucagon-like peptide-1 related to administration relative to meal intake in subjects with type 2 diabetes. J Intern Med. 2001 Jul;250(1):81-87
What is the optimum duration of GLP-1 therapy during a 24 h period? Subjects with type 2 diabetes (considered to have failed SU therapy) were randomized to receive continuous subcutaneous GLP-1 infusion for either 16 (8 am-12 am) or 24 h. Patients treated with 24 h GLP-1 infusions exhibited lower glycemic excursion profiles during the night, and lower fasting blood glucose. These findings suggest that maintaining enhanced levels of GLP-1 signaling even during the overnight period is important for optimal glycemic control in Type 2 diabetes. See Glucagon-Like Peptide-1 Infusion Must Be Maintained for 24 h/day to Obtain Acceptable Glycemia in Type 2 Diabetic Patients Who Are Poorly Controlled on Sulphonylurea Treatment. Diabetes Care. 2001 Aug; 24(8): 1416-21
See GLP-1 action in human subjects
What are the key determinants of GLP-1 action in human subjects with type 2 diabetes? Among the factors to be considered are the route of administration and engagement of enteric neural or portal glucose sensors, and the potential for genetic variation in the GLP1R or acquired forms of GLP-1 resistance to modify patient outcomes in people treated withGLP-1 R agonists. Overgaard and colleages examined a range of parameters that might influence GLP-1 outcomes in people treated with oral vs. injectable semaglutide therapy in the PIONEER vs. the SUSTAIN trials. Remarkably, the circulating levcels of semaglutide, independent of thr route of administration, were the key determinants of semaglutide outcomes
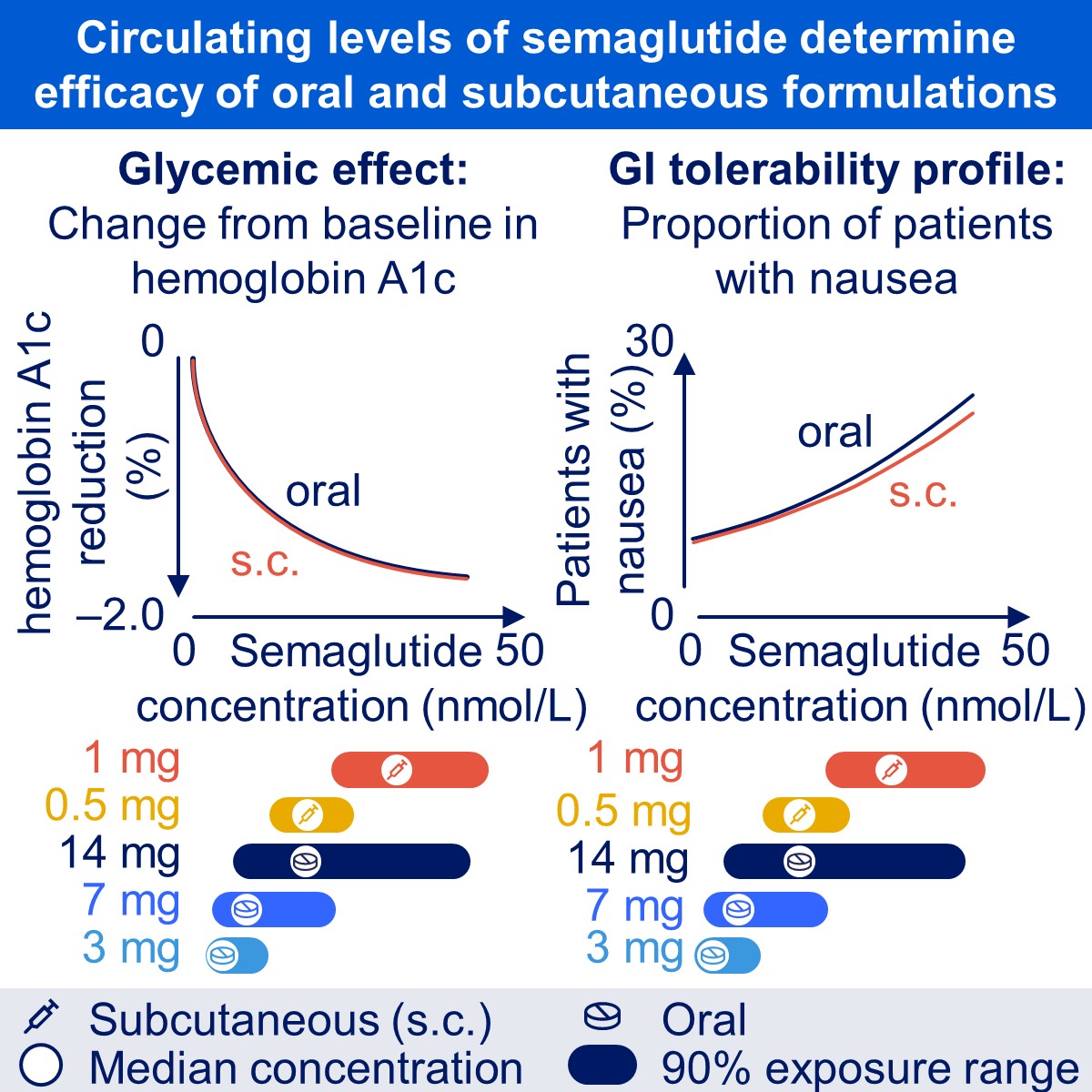
Overgaard, R.V., .Hertz, C. L., Ingwersen, S. H., Navarria, A., Drucker, D.J. Levels of circulating semaglutide determine reductions in HbA1c and body weight in people with type 2 diabetes Cell Reports Medicine 2021 Sept 3 https://doi.org/10.1016/j.xcrm.2021.100387
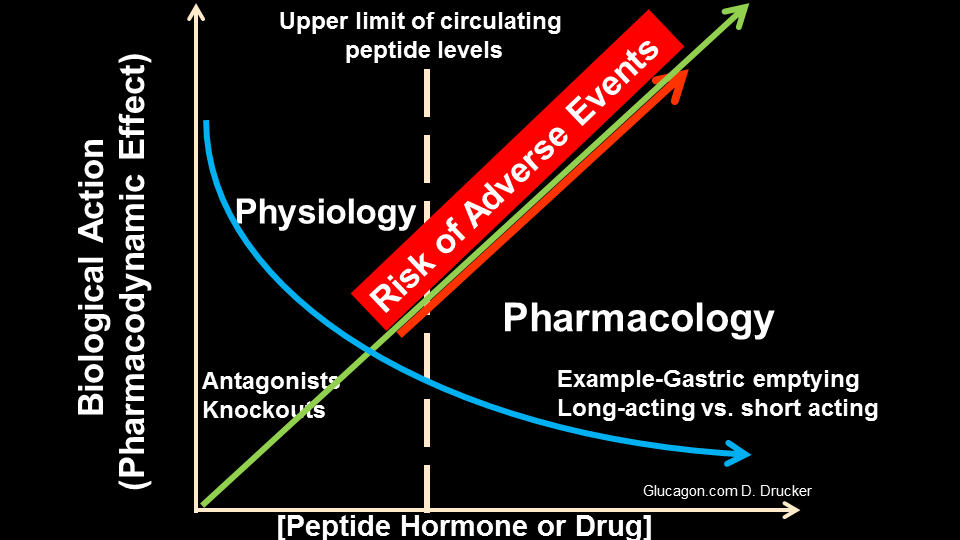
The challenge in developing GLP-1-based therapeutics encompasses delivery of sufficient amounts of bioactive drug, through a titration regimen that minimizes gastrointestinal adverse events.