The Therapeutic Actions of GLP-2 in Human Subjects
GLP-2 exerts potent trophic and anti-apoptotic effects in the GI tract of rodents and enhances nutrient absorption in mice and rats following small bowel resection. Does GLP-2 exert the same effects in human patients with short bowel syndrome? Review the data in the first human study "GLP-2 Improves Nutrient Absorption and Nutritional Status in Short-Bowel Patients With No Colon" in the March 2001 issue of Gastroenterology, and the accompanying Editorial.
The European Medicines Authority recommended approval of a degradation-resistant GLP-2 analogue, Teduglutide for the treatment of short bowel syndrome, on June 22 2012. The United States Food and Drug Administration approved Teduglutide (Gattex) on December 21 2012. Review the Teduglutide Prescribing Information
h[Gly2]GLP-2 (Teduglutide) has been developed by NPS Pharmaceuticals as a potential therapeutic agent for human subjects with intestinal diseases characterized by insufficient repair of intestinal mucosa and/or compromised nutrient absorption.
Human subjects (age 18 or greater) with short bowel syndrome (SBS) rquiring TPN for a minimum of 3 days per week, for at least the past 12 months, were randomized to treatment with either teduglutide (n=43), 0.05 mg/kg/day, or placebo (n=43) for a total of 24 weeks, followed by an ongoing extension phase. Patients were recruited from 27 sites in 10 countries across North America and Europe. Patients were randomized after an initial optimization of up to 8 weeks to stabilize urine output in the 1-2 Liters/day range, to minimize the risk of dehydration. After 24 weeks of therapy, there was a significant reduction in parenteral nutrition requirements (4.4 vs 2.3 L), days off TPN, and an increase in plasma citrulline in teduglutide-treated patients. Oral fluid intake was significantly higher in subjects treated with placebo. Responder rates were higher for subjects with a colon in continuity. Adverse event reports were similar in the 2 groups, with abdominal discomfort, nausea, stomal irritation and cholecystitis reported in teduglutide-treated subjects. Six patients developed non-neutralizing anti-teduglutide antibodies with no clinical sequelae. Teduglutide Reduces Need for Parenteral Support Among Patients with Short Bowel Syndrome with Intestinal Failure Gastroenterology. 2012 Sep 11. pii: S0016-5085(12)01316-9
A 24 week randomized placebo-controlled Phase 3 study of h[Gly2]GLP-2 was carried out in 83 patients with SBS receiving once daily dosing of 0.05 (n=35) or 0.1 mg/kg/day (n=32) or placebo. The primary study endoint was a 20% reduction in parenteral nutrition requirements at week 20 and 24. The algorithm used for weaning patients allowed for a 10% reduction in PN volumes every 4 weeks, guided by changes in urinary output. A maximum of 5 reductions of 10% were permitted over a 24 week period. The low dose, but not the high dose cohort, was associated with a statistically significant reduction in TPN requirements at the end of the study. Surrogate biomarkers for GLP-2 action such as intestinal villus height, plasma citrulline and lean body mass were significantly increased in patients treated with h[Gly2]GLP-2. Three subjects came off TPN, 2 in the low dose arm after 25 and 6.5 years of TPN, and one patient who was TPN dependent for 3.7 yrs came off TPN in the high dose arm. The most common AEs on h[Gly2]GLP-2 were abdominal pain (24%), headache (24%), nausea (22%), nasopharyngitis (16%) and vomiting (15%). Randomised placebo-controlled trial of teduglutide in reducing parenteral nutrition and/or intravenous fluid requirements in patients with short bowel syndrome Gut. 2011 Feb 11. [Epub ahead of print]
In a 21 day pilot study of Teduglutide, in 16 human subjects with short bowel syndrome, with and without a colon in continuity. Teduglutide improved energy absorption, in association with expansion of mucosal crypt plus villus height in the small bowel, as outlined in Teduglutide (ALX-0600), a dipeptidyl peptidase IV resistant glucagon-like peptide 2 analogue, improves intestinal function in short bowel syndrome patients. Gut. 2005 Sep;54(9):1224-31
Analysis of bone density in eight short bowel patients treated with synthetic human GLP-2, 400 ug sc twice daily for 5 weeks demonstrated significant increases in spinal bone mineral density in 7/8 patients, with an increase in ionized calcium detected in 5/8 patients. The mechanism for these findings remains unclear, but may be due in part to increased calcium absorption. See Short-term administration of glucagon-like peptide-2. Effects on bone mineral density and markers of bone turnover in short-bowel patients with no colon. Scand J Gastroenterol. 2002 Apr;37(4):392-8.
Longer follow up in a small number of patients treated with native GLP-2 reported modest but significant sustained effects on energy absorption, and a significant reduction of fecal wet weight by ~1 kg/day, enabling patients to maintain their fluid and electrolyte status with reduced oral intake, with a modest improvement in kidney function Short bowel patients treated for two years with glucagon-like Peptide 2: effects on intestinal morphology and absorption, renal function, bone and body composition, and muscle function. Gastroenterol Res Pract. 2009:616054. Epub 2009 Aug 20.
GLP-2 has been shown to exhibit promising therapeutic potential in experimental models of intestinal atrophy and injury including:
TPN-induced mucosal atrophy
Prevention of parenteral nutrition-induced gut hypoplasia by coinfusion of glucagon-like peptide-2. Am J Physiol. 1997;273 :G559-63 Intravenous GLP-2 reverses the TPN-associated mucosal villus hypoplasia in the small bowelMaintaining gut integrity during parenteral nutrition of tumor-bearing rats: effects of glucagon-like peptide 2. Nutr Cancer. 2000;37(2):215-22 GLP-2 infusion reverses mucosal atrophy in the small bowel without effects on tumor growth in tumor-bearing ratsShort bowel syndrome following major intestinal resection
GLP-2 augments the adaptive response to massive intestinal resection in rat. Am J Physiol. 1998 Nov;275(5 Pt 1):G911-21 GLP-2 improves nutrient absorption and augments intestinal adaptation in rats following MSBR. Consistent with a role for endogenous GLP-2 in intestinal adaptation, the levels of circulating GLP-2 are rapidly and significantly elevated in the rat following MSBR. See Time-dependent intestinal adaptation and GLP-2 alterations after small bowel resection in rats. Am J Physiol Gastrointest Liver Physiol. 2001 Sep;281(3):G779-85. The increase in endogenous levels of GLP-2 after major small bowel resection appears to be an important component of intestinal adaptation as rats administered GLP-2 antisera exhibited diminished histological features of intestinal adaptation in the remnant ileum as shown in Glucagon-like Peptide 2 is an endogenous mediator of postresection intestinal adaptation. JPEN J Parenter Enteral Nutr. 2005 Mar-Apr;29(2):97-101
The timing of optimal GLP-2 administration was assessed in mice following ileo-cecal resection. Groups of mice were treated with GLP-2 immediately after ileo-cecal resection, or commencing 5 weeks after surgery. Only mice receiving early GLP-2 therapy exhibited therapeutic benefit as measured by augmentation of intestinal progenitors, increased cell proliferation and crypt fission, and more rapid attainment of increased mucosal circumference as outlined in Early but not late administration of glucagon-like peptide-2 following ileo-cecal resection augments putative intestinal stem cell expansion Am J Physiol Gastrointest Liver Physiol 2009;296 G643-G650.
Experimental colitis in mice
Human [Gly2]GLP-2 reduces the severity of colonic injury in a murine model of experimental colitis Am J Physiol 1999 Jan;276(1 Pt 1):G79-91 GLP-2 markedly attenuates weight loss and reduces multiple histological parameters of intestinal injury in the colon of mice with DS colitis as also described in Glucagon-like peptide-2 and common therapeutics in a murine model of ulcerative colitis. J Pharmacol Exp Ther. 2003 Jul;306(1):347-54. Similarly, GLP-2 does not require IL-10 for its anti-inflammatory and therapeutic actions as administration of GLP-2 (50 microg/kg/sc. injection) treatment for 5 days to IL-10-/- mice produced significant amelioration of disease activity, in association with an increase in intestinal epithelial cell SOCS-3 expression and reduced STAT-3 signaling as outlined in Interleukin-10 independent anti-inflammatory actions of glucagon-like peptide 2. Am J Physiol Gastrointest Liver Physiol. 2008 Oct 9. GLP-2 also reduces the severity of colitis in rats either following injection of TNBS, or after oral administration of DS, actions which appear to require VIP, as described in Enteric neural pathways mediate the anti-inflammatory actions of glucagon-like peptide 2. Am J Physiol Gastrointest Liver Physiol. 2007 Jul;293(1):G211-21.
Sigalet and colleagues further examined the interaction between GLP-2 and inflammation in the rat model of TNBS-induced colitis by examining the effect of GLP-2 on enteric neuronal populations. a 5 day treatment course with GLP-2 (twice daily) reduced neutrophil infiltration, reduced histological parameters of colonic injury, reversed the loss of enteric neurons, with a selective increase in the number of VIP+ but not nNOS+ cells. Intriguingly, GLP-2 treatment also increased the number of VIP+ cells in the absence of inflammation. See The effects of glucagon-like peptide 2 on enteric neurons in intestinal inflammation Neurogastroenterol Motil. 2010 Aug 16
Non-steroidal anti-inflammatory drug-induced enteritis in mice and rats
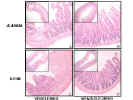 Administration of GLP-2 markedly decreased NSAID-induced
intestinal in mice, as shown below
Administration of GLP-2 markedly decreased NSAID-induced
intestinal in mice, as shown below
Glucagon-like peptide 2 decreases mortality and reduces the severity of indomethacin-induced murine enteritis Am J Physiol 1999 Nov;277(5 Pt 1):E937-47 GLP-2, administered in a variety of regimens, reduces mortality and improves gross and microscopic indices of intestinal injury in mice with NSAID-induced enteritis
GLP-2 administration in experimental vascular ischemia
Evidence for the protective effects of GLP-2 in the setting of experimental intestinal injury is described in Glucagonlike peptide-2 analogue enhances intestinal mucosal mass after ischemia and reperfusion J Pediatr Surg. 2000 Feb;35(2):357-9. This study shows that GLP-2 promotes mucosal repair and significantly reduces mortality in rats following experimental SMA occlusion. Similarly, GLP-2 was administered prior to ischemia-reperfusion for 3 days, or prior to and following I/R, in rats. The authors concluded that treatment with GLP-2 attenuates intestinal I/R injury, reduces bacterial translocation, inhibits the release of oxygen free radicals and ET-1, and inhibitd the production of proinflammatory cytokines Protective effects of glucagon-like peptide 2 on intestinal ischemia-reperfusion rats Microsurgery. 2008 Mar 27;28(4):285-290. [Epub ahead of print]
GLP-2 and Immune-mediated IBD in rats
Treatment of inflammatory bowel disease in a rodent model with the intestinal growth factor glucagon-like peptide-2 J Pediatr Surg 2000 Jun;35(6):847-51 Administration of GLP-2 as a 14 day intravenous infusion at 50 microg/kg/d significantly reduced gross and histological intestinal mucosal damage and reduced the levels of the cytokines TNF-alpha and IFN-gamma in rats with antigen induced enteritis.
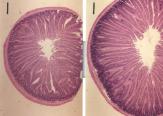 Increased levels of GLP-2 likely mediate small bowel villus
hyperplasia in experimental diabetes
Increased levels of GLP-2 likely mediate small bowel villus
hyperplasia in experimental diabetes
Treatment of rats with insulin reverses the villus hyperplasia (left panel compared to right) and reduces circulating levels of GLP-2 See Intestinal growth is associated with elevated levels of glucagon-like peptide 2 in diabetic rats. Am J Physiol 1997 273: E815-20
GLP-2 effects in premature pigs
GLP-2 administration enhanced DNA synthesis, increased villus height, reduced apoptosis and reduced proteolysis in the developing pig gut. See GLP-2 stimulates intestinal growth in premature TPN-fed pigs by suppressing proteolysis and apoptosis AJP Vol. 279, Issue 6, G1249-G1256, December 2000
Administration of human GLP-2 by continuous infusion for 48 hrs prior to enteral nutrient administration in TPN-fed piglets (born by C-section 12 days premature) and up to 96 h after initiation of enteral administration was used to assess whether GLP-2 could prevent or ameliorate signs of gut injury in a pig model of necrotizing enterocolitis. The initial manifesttaions of NEC were significantly delayed in GLP-2-infused pigs, and gut mucosal histology was better preserved in the jejunum of GLP-2-treated piglets. However final mortality was not different, plasma VIP levels were comparable across groups, and gut inflammation (myeloperoxidase) and histology was not different and serum amyloid A levels were higher in pigs treated with GLP-2. GLP-2 Delays But Does Not Prevent the Onset of Necrotizing Enterocolitis in Preterm Pigs J Pediatr Gastroenterol Nutr. 2013 Jan 21. Once daily administration of teduglutide for 7 days in newborn TPN-fed pigs following 50% jejujunal resection increased small bowel growth but had minimal effect on digestive enzyme expression or nutrient absorption Acute Effects of the Glucagon-Like Peptide 2 Analogue, Teduglutide, On Intestinal Adaptation in Newborn Pigs With Short Bowel Syndrome J Pediatr Gastroenterol Nutr. 2014 Jan 3
GLP-2 and cancer
Does GLP-2 exert its intestinotrophic actions in the setting of neoplasia? Treatment of tumor-bearing rats with TPN alone, versus TPN plus co-administered GLP-2 demonstrated that the trophic effects of GLP-2 on the small intestinal mucosa were maintained in tumor-bearing rats. In contrast, no significant effects of GLP-2 were noted in the colon. GLP-2 did not reduce tumor-bearing immunosuppression, and GLP-2 had no effect on tumor growth, whereas bacterial translocation was modestly reduced in GLP-2-treated animals. See Maintaining gut integrity during parenteral nutrition of tumor-bearing rats: effects of glucagon-like peptide 2. Nutr Cancer. 2000;37(2):215-22
In contrast, studies in mice administered the cancer inducing methylating carcinogen 1,2-dimethylhydrazine (DMH) demonstrated that concomitant GLP-2 administration for 10 or 30 days increased the number of colonic polyps, although no effect of GLP-2 treatment on survival was detected, See Glucagon-like peptide 2 (GLP-2) accelerates the growth of colonic neoplasms in mice. Gut. 2004 Aug;53 (8): 1145-50. A subsequent study demonstrated that twice daily administration of 1.5 ug [Gly2]-GLP-2 for 4 weeks to mice previously treated with azoxymethane resulted in an increased number of aberrant crypt foci and two GLP-2-treated animals developed colonic adenocrcinomas Carcinogenic effects of exogenous and endogenous glucagon-like peptide-2 in azoxymethane-treated mice. Endocrinology. 2009 Sep;150(9):4033-43. Epub 2009 Jun 4.
GLP-2 and chemotherapy
 GLP-2 administration prevents chemotherapy-induced apoptosis. How does GLP-2 receptor activation in the gut endocrine cell reduce apoptosis in the
gastrointestinal epithelium, leading to increased survival in experimental
chemotherapy- induced mucositis? Review the data in Glucagon-like
Peptide (GLP)-2 Reduces Chemotherapy-associated Mortality and Enhances
Cell Survival in Cells Expressing a Transfected GLP-2 Receptor Cancer Res 2001 61: 687-693
GLP-2 administration prevents chemotherapy-induced apoptosis. How does GLP-2 receptor activation in the gut endocrine cell reduce apoptosis in the
gastrointestinal epithelium, leading to increased survival in experimental
chemotherapy- induced mucositis? Review the data in Glucagon-like
Peptide (GLP)-2 Reduces Chemotherapy-associated Mortality and Enhances
Cell Survival in Cells Expressing a Transfected GLP-2 Receptor Cancer Res 2001 61: 687-693
A similar correlation between increased levels of GLP-2 and protection from chemotherapy-induced enteritis has been demonstrated in mice treated with 5-FU and either GLP-2 alone or both metformin and Val-Pyr, which enhances the levels of circulating GLP-2. See The combination of metformin and a dipeptidyl peptidase IV inhibitor prevents 5-fluorouracil-induced reduction of small intestine weight. Eur J Pharmacol. 2004 Mar 19;488(1-3):213-8
GLP-2 AND DIABETES
Experimental diabetes has been associated with intestinal mucosal hyperplasia in rats and mice Structural and functional studies on the transformation of the intestinal mucosa in rats with experimental diabetes Res Exp Med (Berl). 1977 Jun 29;170(2):89-99 and Mechanisms of adaptation in rat small intestine: regional differences in quantitative morphology during normal growth and experimental hypertrophy J Anat. 1989 Jun;164:189-200 , and this might be due in part to hyperphagia or increased expression of gut growth factor signaling, perhaps related to abnormalities in insulin action. GLP-2 has been postulated to play a causal role in the devlopment of gut growth associated with diabetes in rodents. Circulating and ileal tissue levels of GLI-peptides were significantly elevated in the STZ-diabetic rat as early as 8 days following induction of diabetes Tissue-specific differences in the levels of proglucagon-derived peptides in streptozotocin-induced diabetes Endocrinology. 1989 Jun;124(6):3003-9. Circulating levels of GLP-1, GLP-2 and GLI-peptides were also significantly increased in STZ-diabetic rats 3 weeks after induction of diabetes with a parallel increase in intestinal growth as measured by small intestinal wet and dry weight, and crypt+villus height Intestinal growth is associated with elevated levels of glucagon-like peptide 2 in diabetic rats Am J Physiol. 1997 Oct;273(4 Pt 1):E815-20. Immunoneutralization of circulating GLP-2 using specific polyclonal antisera raised against synthetic human GLP-2 in rabbits attenuated the STZ-induced increase in intestinal growth in the rat (35% increase in mucosal area vs. 66% increase in mucosal area in mice treated with non-specific antibodies), suggesting that endogenous GLP-2is required for the increased diabetic intestinal growth Immunoneutralization of endogenous glucagon-like peptide-2 reduces adaptive intestinal growth in diabetic rats Regul Pept. 2002 May 30;105(3):173-9. In contrast, induction of diabetes using acute high dose STZ in Glp2r-/- mice increased small intestinal weight to the same extent inGlp2r-/- vs.Glp2r+/+ mice. These results suggest endogenous GLP-2R signaling is not required for diabetic intestinal growth in mice Glucagon-like peptide-2 receptor modulates islet adaptation to metabolic stress in the ob/ob mouse Gastroenterology. 2010 Sep;139(3):857-68
Obese human subjects with chronic hyperglycemia were also found to have increased small bowel enterocyte loss (assessed by measurement of plasma citrulline), however fasting plasma levels of GLP-2 were only modestly increased (14 vs 18 ng/ml for A1c less than vs greater than 6.0) in these subjects and did not correlate with estimates of enterocyte mass. Small Intestinal Alterations in Severely Obese Hyperglycemic Subjects J Clin Endocrinol Metab. 2010 Nov 17. [Epub ahead of print]
GLP-2 in experimental burn injury
GLP-2 infusion in burned rats prevented loss of gut protein and enhanced immune function. Glucagon-like peptide-2 stimulates gut mucosal growth and immune response in burned rats. J Burn Care Rehabil. 2001 Mar-Apr; 22(2):136-43.
GLP-2 in acute necrotizing pancreatitis
The effects of a GLP-2 analog, ALX-0600, have been examined in the setting of experimental necrotizing pancreatitis induced in Sprague-Dawley rats. Treatment with ALX-0600 for 3 days significantly reduced intestinal permeability, with a trend to reduction of bacterial translocation in viscera and mesenteric lymph nodes. See The effect of glucagon-like peptide 2 on intestinal permeability and bacterial translocation in acute necrotizing pancreatitis. Am J Surg. 2001 Jun;181(6):571-5.
GLP-2 in acute radiation-induced injury
