The most common side effects or adverse events associated with GLP-1 therapy is nausea, and occasionally vomiting, followed by diarrhea/constipation. For the majority of subjects, the extent of nausea generally diminishes over time, and most subjects do not report ongoing nausea after several months of therapy with a GLP-1R agonist. Diarrhea has also been reported in subjects taking GLP-1R agonists. Other reported AEs may include hypoglycemia, particularly if a GLP-1R agonist is used together with an insulin secretagogue, such as a sulfonylurea. Injection site reactions, including itching and skin rashes have also been described. Less commonly, allergic reactions have been reported. An enormous amount of literature, describing both preclinical clinical studies, has addressed the topic of incretin-based safety. Some of the data is summarized below.
The United States Food and Drug Administration announced March 14 2013 it is investigating reports of pancreatitis and unpublished preclinical findings related to potential precancerous findings in studies of the pancreas using incretin-based therapies. The European Medicines Agency also investigated, in the spring and summer of 2013, the link between GLP-1 action and possible development of pancreatitis and cancer. EMEA concluded in July 2013 that " In conclusion, the results of the study by Butler et al are not considered to constitute a new safety signal for the GLP 1 based therapies with respect to pancreatic safety. This is further supported by the review of available preclinical and clinical data". The Assessment report for GLP-1 based therapies is a public document. Moreover, in a joint summary paper published in the NEJM on February 27 2014, authors from bth EMEA and FDA summarized independent reviews of data from academic labs, the FDAs own analyses, and review of all relevant clinical trial data. The authors stated that:
Both agencies agree that assertions concerning a causal association between incretin-based drugs and pancreatitis or pancreatic cancer, as expressed recently in the scientific literature and in the media, are inconsistent with the current data. FDA and EMA have not reached a final conclusion at this time regarding such a causal relationship. Although the totality of the data that have been reviewed provided reassurance, pancreatitis will continue to be considered a risk associated with these drugs until more data are available. FDA and EMA believe that the current knowledge is adequately reflected in the product information or labeling, and further harmonisation among products is planned in Europe
Many of the studies that have informed our understanding of incretin safety, and in some cases, generated considerable debate surrounding scientific veracity, are summarized below.
Pancreatitis and Pancreatic cancer
Clinical perspectives
To review the May 2007 AGA criteria for diagnosis and management of acute pancreatitis, see AGA Institute medical position statement on acute pancreatitis. The diagnosis of acute pancreatitis in subjects with diabetes may be more challenging than in non-diabetic subjects due to a much greater proportion of asymptomatic diabetic subjects that exhibit increased circulating levels of lipase and amylase, without other classic symptoms or signs or imaging abnormalities associated with pancreatitis. Increased variability and abnormalities in pancreatic enzyme concentrations in otherwise asymptomatic subjects with type 2 diabetes Diabetes Metab Syndr Obes. 2012;5:419-24
The vast majority of epidemiologic studies assessing possible links between GLP-1R agonists and DPP-4 inhibitors have been retrospective database analyses, which have multiple scientific limitations. Meier & Nauck reported a meta-analyses for the available data describing pancreatitis events in phase 3 trials for DPP-4 inhibitors and GLP-1R agonists. The odds ratio for acute pancreatitis with a GLP-1R agonist was 1.39, whereas for exposure to a DPP-4 inhibitor, it was 1.07, however the total number of patients studied and absolute event rates were small (2.1 events of pancreatitis per 1,000 patient years), greater numbers of subjects followed for longer periods of time would be required to increase the statistical power of the conclusions. Risk of pancreatitis in patients treated with incretin-based therapies Diabetologia. 2014 Apr 11.
Pancreatitis rates have been reported in subjects randomized to either saxagliptin (SAVOR TIMI53) or alogliptin (EXAMINE) in cardiovascular outcome studies. Pancreatitis events were independently adjudicated. The number of events of all pancreatitis (acute and chronic) were similar in saxagliptin vs. placebo-treated subjects (mean follow up period of 2.1 years, 16,884 person-years in the saxagliptin
group and 16,761 person-years in placebo group ). Although slightly more events of acute pancreatitis and fewer cases of chronic pancreatitis were reported in subjects randomized to saxagliptin, the event rates were low, and the small differences did not reach statistical significance. Between 80-90% of patients with pancreatitis had other classical predisposing risk factors and the time to onset of pancreatitis was not different across treatment groups. Fewer events of pancreatic cancer were reported in subjects randomized to saxagliptin. Incidence of Pancreatitis and Pancreatic Cancer in a Randomized Controlled Multicenter Trial (SAVOR-TIMI 53) of the Dipeptidyl Peptidase-4 (DPP-4) Inhibitor Saxagliptin Diabetes Care. 2014 Jun 9. pii: DC_132546
Adverse event reporting has raised an association between use of Exenatide and pancreatititis, an inflammation of the pancreas that can, in rare cases be quite severe, and even fatal. There is limited information about the true incidence of pancreatitis in patients with diabetes but most studies consistently show ~2-fold increased rate pf panceatitis in diabetic subjects. A retrospective claims database analysis from Amylin Pharmaceuticals Inc (now BMS) suggested that patients with diabetes have a 2.8-fold greater risk of developing pancreatitis and a 1.9-fold greater risk of developing biliary tract disease; the relative risk for pancreatitis was highest in younger patients les than 45 years of age- See Increased Risk of Acute Pancreatitis and Biliary Disease Observed in Patients with Type 2 Diabetes: a Retrospective, Cohort Study Diabetes Care 2009 May;32(5):834-8 Analysis of hospitalized patients diagnosed with pancreatitis in a large health insurance data base was carried out using data assessed from June 2005-2008. Patients were analyzed by the type of anti-diabetic therapy they were using, with a specific focus on patients taking exenatide vs sitagliptin. Acute pancreatitis ocurred in 0.13% and 0.12% of patients taking exenatide vs. sitagliptin, respectively, rates comparable to patients on metformin/glyburide as outlined in Use of a claims-based active drug safety surveillance system to assess the risk of acute pancreatitis with exenatide or sitagliptin compared to metformin or glyburide Curr Med Res Opin. 2009 Apr;25(4):1019-27.
Adverse events, including pancreatitis and pancreatic cancer, were assessed in a pooled meta-analysis of 25 clinical studies of sitagliptin (7,726 patients on sitagliptin vs. 6,885 non-exposed control subjects) ranging from 12 weeks to 2 years. Mean exposure to drug across the entire pooled analsysi was 284 days; 32% of patients were treated with sitagliptin for at least 1 year, and 8% received sitagliptin for 2 years. The incidence of all reported malignancies, including pancreatic cancer was similar across treatment groups, however too few events were reported to calculate 95% confidence intervals. Angioedema was not more common after exposure to sitagliptin, and there was no difference in reported rates of pancreatitis. Rash was more commonly reported in sitagliptin-treated subjects Safety and Tolerability of Sitagliptin in Type 2 Diabetes: Pooled Analysis of 25 Clinical Studies Diabetes Ther. 2013 May 23
Wenten and colleagues assessed the relative risk of pancreatitis using a health care claims database in 482,034 eligible patients who had started a new medication for the treatment of type 2 diabetes; 24,237 initiated exenatide twice daily and 457,797 initiated another anti-diabetic medication. Patients starting exenatide were felt to have more advanced forms of diabetes. No significant association was found between exenatide use and the development of diabetes. Relative risk of acute pancreatitis in initiators of exenatide twice daily compared with other anti-diabetic medication: a follow-up study. Diabet Med. 2012 Nov;29(11):1412-8
Singh et al analyzed pooled health care claims administrative database data for cases of acute pancreatitis for diabetic subjects with administrative codes suggestive of inpatient pancreatitis, and compared rates of multiple variables, including exenatide or sitagliptin use, to rates reported for randomly selected controls. Pancreatitis reporting rates were ~2.2-fold higher among recent users (prescriptions written, use not verified) of exenatide or sitagliptin. The duration of diabetes for cases vs. control was not reported. The authors chose not to report relative pancreatitis rates for patients treated with any other diabetes therapy (insulin, metformin, SUs, TZDs etc). Glucagonlike Peptide 1–Based Therapies and Risk of Hospitalization for Acute Pancreatitis in Type 2 Diabetes Mellitus JAMA Intern Med. 2013;():1-6. doi:10.1001/jamainternmed.2013.2720
Case reports of pancreatitis have also been described, and the FDA has provided updates through correspondence in the literature and periodic safety alerts, as outlined in FDA NEJM Pancreatitis and Information for healthcare professionals about Exenatide and pancreatitis .
Elashoff and colleagues reviewed case reports of adverse events submitted to the Adverse Event Reporting System Database from Q1 2004 through Q2 2009 with the aim of detecting cases of a) pancreatitis b) thyroid cancer and pancreatic cancer and c) all cancers linked to associated use of sitagliptin (DPP-4 inhibitor) or exenatide (GLP-1R agonist) for the treatment of type 2 diabetes. A brief FDA AERS overview provides independent information about the utility of the AERS database. Control rates were ascertained by Elashoff by querying rates for cancer, pancreatitis, and other pre-defined events that may or may not be associated with use of other anti-diabetic drugs; rosiglitazone (Avandia), nateglinide (Starlix), repaglinide (Prandin), and glipizide. Control events that were also queried for use in this analysis were back pain, urinary tract infection, chest pain, cough, and syncope. The authors discarded the use of pioglitazone from their AERS analysis, since it was noted to be associated with an elevated reporting rates for control events not known to be associated with pioglitazone in other database analyses. Reported rates of pancreatitis were six-fold higher for exenatide and sitagliptin for pancreatitis and reported event rates in this analysis for pancreatic cancer was noted to be 2.9-fold greater with exenatide and 2.4-fold greater with sitagliptin. More thyroid cancer was reported in patients using exenatide and case report rates for "all other cancers" were higher with sitagliptin. See Increased Incidence of Pancreatitis and Cancer Among Patients Given Glucagon Like Peptide-1 Based Therapy Gastroenterology. 2011 Jul;141(1):150-6. A number of epidemiological surveys and data base analyses reveal an increased incidence of multiple neoplasms, including pancreatic cancer in subjects with T2DM Association between diagnosed diabetes and self-reported cancer among U.S. adults: findings from the 2009 Behavioral Risk Factor Surveillance System Diabetes Care. 2011 Jun;34(6):1365-8
A retrospective case control study of 268,561 subjects with diabetes revealed that over a several year period (2007-2009), ~2.6% of subjects reported use of exenatide. Overall, hospitalization rates for acute pancreatitis were higher in subjects who did not use exenatide. A similar analysis of claims for pancreatic cancer revealed no significant association between exenatide use and pancreatic cancer diagnosis Exenatide therapy and the risk of pancreatitis and pancreatic cancer in a privately insured population Diabetes Technol Ther. 2012 Oct;14(10):904-11
An overview of the safety issues and current understanding of the risk:benefit ratio for the use of GLP-1R agonists and DPP-4 inhibitors was published in the July 2011 issue of JCEM The safety of incretin-based therapies; review of the scientific evidence J Clin Endocrinol Metab 2011 96(7);2027-2031
A meta-analysis of serious adverse events reported during evaluation of clinical trials of at least 24 weeks duration for DPP-4 inhibitors (sitagliptin, saxagliptin, linagliptin, vildaglitpin, dutogliptin) focused on reporting of cancers, pancreatitis, and cardiovascular (MACE) events. No signal was reported for cancer or pancreatitis, whereas MACE events were relatively reduced in subjects exposed to DPP-4 inhibitors, however the short term nature of these studies precludes definitive conclusions. Safety of dipeptidyl peptidase-4 inhibitors: a meta-analysis of randomized clinical trials Curr Med Res Opin. 2011 Nov;27 Suppl 3:57-64
Similarly, an independent meta-anlysis of 25 clinical trials using the GLP-1R agonists exenatide or liraglutide found no conclusive evidence that the use of these agents was associated with a significantly increased risk of acute pancreatitis or cancer, compared to active comparators in the same trials A meta-analysis of serious adverse events reported with exenatide and liraglutide: acute pancreatitis and cancer Diabetes Res Clin Pract. 2012 Nov;98(2):271-84
Butler and colleagues reported histopathological findings in pancreata obtained from organ donors with diabetes treated with (8) or without incretin-based therapy (12) vs. findings in non-diabetic controls (12). All subjects had been treated with incretin-based therapy (sitagliptin (7) or exenatide (1) for at least a year, however the duration of exposure was not provided, and the interval between comencement of life support, death, procurement of the pancreas, and last exposure to an incretin agent was not described. There were substantial differences in regard to age (58), sex (6 M, 2F) and duration of diabetes (12) for pancreata from subjects exposed to incretins, vs control DM groups (4M, 8F), age (35) and 8 years of previous diabetes. All of the incretin-treated subjects but one were on 2 or more anti-diabetic medications whereas 5 DM controls were taking no anti-diabetic medications and 6 were treated with only 1 agent. Three of the control subjects (2 described in the paper, one in the database) yet none of the incretin-treated subjects had a history of diabetic ketoacidosis. The beta cell mass and pancreatic weight and age for the control diabetic subjects was generally much lower than for the diabetic pancreases from subjects treated with incretin therapy. Pancreatic intraepithelial neoplasia (PanIN) was scored histologically by a pathologist blinded to the clinical data. Donor subjects treated with incretin therapy exhibited an increase in pancreatic mass of 40%; Beta cell mass was decreased by 55% in diabetic control subjects, whereas incretin-treated subjects exhibited a 6-fold increase in beta cell mass relative to diabetic controls and a 3-fold increase in beta cell mass vs. non-diabetic controls. This increase was largely secondary to an increase in beta cell number rather than size. The pancreatic fractional area immunopositive for glucagon was increased ~2.8 fold in subjects treated with incretins, largely due to increased numbers of islet alpha cells. Enlarged islets andincreased numbers of insulin-positive and glucagon-positive cells adjacent to duct structures were more commonly observed in pancreatic from donor subjects treated with incretins. The single pancreas from an exenatide-treated individual exhibited marked alpha cell hyperplasia. A 1.5 cm glucagon-producing alpha cell tumor was detected in the pancreas from a subject treated with sitagliptin; glucagon-producing microadenomas were also obsrved in 2 additional subjects and hyperplastic glucagon-immunopositive islets were detected in 7/8 pancreases from subjects that received incretin therapy. More islet cells exhibiting dual immunopositivity for both insulin and glucagon were detected in pancreata from incretin-treated subjects. Pancreatic intraepithelial neoplasia lesions (low grades 1&2, not broken out separately) were more common in pancreata exposed to incretins. Although the authors repeatedly speculate that reduced glucagon secretion arising secondary to incretin therapy likely accounts for the alpha cell hyperplasia they observed, they did not provide a reference linking partial reduction of glucagon secretion to the development of alpha cell hyperplasia. Marked Expansion of Exocrine and Endocrine Pancreas with Incretin Therapy in Humans with increased Exocrine Pancreas Dysplasia and the potential for Glucagon-producing Neuroendocrine Tumors Diabetes published ahead of print March 22, 2013, doi:10.2337/db12-1686. A commentary on this paper appeared online April 17 2013, raising many similar issues in regard to challenges in the interpretation of the data reported Incretin Therapy and Islet Pathology – A Time for Caution Diabetes published ahead of print April 17, 2013, doi:10.2337/db13-0520
Extensive critical appraisal of the Butler pathology paper has been published by independent research groups. Harja and colleagues noted extensive differences between cases and controls, including differences in age of onset of diabetes, age of subjects analyzed, sex of subjects, inclusion of subjects with T1D (ketoacidsos, autoantibodies), missing data for pancreatic weight, and failure to note abnormalities of glucagon staining in control pancreata An analysis of characteristics of subjects examined for incretin effects on pancreatic pathology. Diabetes Technolo Therap 2013 Aug;15(8):609-18 Bonner-Weir and colleagues re-examined the same pathological slides, where available, and raise multiple concerns about the interpretation of the data. In addition to the multiple significant differences between "controls" vs. cases noted above, they also highlight major methodological issues that challenge the validitiy of the data, including large variability in the intensity of chromogen Fast Red staining and problems with the methodology used to calculate endocrine cell area via hematoxylin staining. They note that the difference in pancreatic weight amongst groups is driven by one extreme outlier with a huge pancreas of 204 g, a measurement more than 2 standard deviations from the mean and a finding approximated by only one of 1238 pancreases donated for islet isolation in the Brussels database. As noted by Harja, the authors question the findings of alpha cell hyperplasia and they note that alpha cell hyperplasia and glucagon+ microaadenomas may be common finding in control pancreata, increasingly observed in older pancreas specimens. Similarly, they note concerns with failure to correct for age in the analysis of PAnIn lesions. They conclude that most of the major conclusions made are not supported by the data and no meaningful conclusions can be drawn from the existing slides. Re-analysis of study of pancreatic effects of incretin therapy: Methodological deficiencies Diabetes Obes Metab. 2014 Jan 8;.
Potentially confounding interpretation of many of the findings reported in this paper, PanIN lesions are known to occur with increasing frequencey in older subjects The prevalence of pancreatic intraepithelial neoplasia in pancreata with uncommon types of primary neoplasms Am J Surg Pathol. 2006 Jan;30(1):36-41 and Pancreatic intraepithelial neoplasms in the normal appearing pancreas: on their precise relationship with age Hepatogastroenterology. 2008 May-Jun;55(84):1103-6. Furthermore, it has been elegantly described previously that a large number of histological changes develop in the endocrine pancreas, inluded large increases in beta cell replication, and increased replication of glucagon-, somatostatin-, and CA19.9-positive cells, as described in pancreata procured from young organ donors subjected to prolonged (more than 3 days) life support ventilation prior to death Beta-cell replication is increased in donor organs from young patients after prolonged life support Diabetes. 2010 Jul;59(7):1702-8. doi: 10.2337/db09-1698. Moreover, the presence of positive anti-islet antibodies in several control diabetic subjects, as well as the likelihood of T1DM in at least 3 of the control diabetic subjects, together with data from the same authors illustrating that pancreatic mass is reduced in subjects with antibodies and further reduced in T1DM subjects, greatly complicates interpretation of the data. Pancreas organ weight in individuals with disease-associated autoantibodies at risk for type 1 diabetes JAMA. 2012 Dec 12;308(22):2337-9.
Does acute or sustained GLP-1 receptor activation modify gallbladder function in diabetic subjects? Keller et al examined gallbladder emptying in normal fasting healthy subjects without a previous history of gallbaldder dysfunction in a randomized double blind crossover study, with administration of exenatide 10ug, or placebo, with and without CCK infusion to stimulate gallbladder emptying. Exenatide significantly reduced GB emptying, with no change in the diameter of the pancreatic or bile ducts. Effect of exenatide on cholecystokinin-induced gallbladder emptying in fasting healthy subjects Regul Pept. 2012 Sep 6.
Waser and colleagues used a combination of in situ ligand autoradiography with 125I-GLP-1(7–36)amide, and immunocytochemistry using a GLP-1R-selective monoclonal antibody, to study GLP-1R expression in normal and neoplastic human pancreas and thyroid. GLP-1R was predominantly localized to islet beta cells and within ductal neuroendocrine cells in the pancreas. GLP-1R immunopositivity was not detected in ductal cells or in 88 pancreatic adenocarcinomas however PanIN 1/2 lesions did exhibit GLPR+ cells (6/20 cases). Low level staining was observed in human acinar cells. GLPR expression was not detected in normal human thyroid, in papillary thyroid cancer, or in 10 cases from MTC related to MEN2. However 2/10 sporadic medullary thyroid cancers did exhibit GLPGLP-1R-immuopositive cells. See Glucagon-like-peptide-1 receptor expression in normal and diseased human thyroid and pancreas Mod Pathol. 2014 Sep 12. doi: 10.1038/modpathol.2014.113
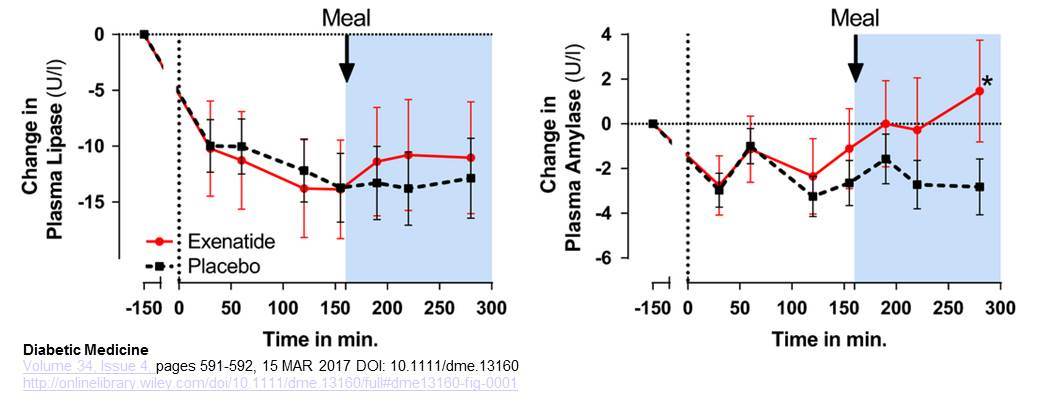
Smits et al examined the profiles of plasma amylase and lipase after acute infusion of exenatide (50 ng/min in 30 min, followed by continuous infusion of 25 ng/min) in human subjects with type 2 diabetes, studied both in the fasting state and following consumption of a meal. Plasma levels of amylase were significantly higher after exenatide, whereas plasma levels of lipase trended higher but were not significantly different within the 280 minute study period. Acute plasma amylase increase after glucagon-like peptide -1 receptor agonist exenatide administration in Type 2 diabetes Diabet Med. 2017 Apr;34(4):591-592
Smits et al reported a number of exocrine pancreas-related outcomes in a small number of patients (n=19)with type 2 diabetes treated with liraglutide or sitagliptin for 12 weeks. Small and transient increases in pancreatic enzymes were deected with both drugs that were not sustained after 12 weeks. Pancreatic intraduodenal fluid secretion was slightly increased with sitagliptin but not liraglutide, and pancreas volume assessed by imaging was not changed. Pancreatic Effects of Liraglutide or Sitagliptin in Overweight Patients With Type 2 Diabetes: A 12-Week Randomized, Placebo-Controlled Trial Diabetes Care. 2016 Dec 20. pii: dc160836. doi: 10.2337/dc16-0836
Preclinical Data
How useful are rodents for studies of pancreatic pathology? Chadwick and colleagues report an extensive analysis of pancreatic lesions detected in three different strains of commonly studied rats, Sprague Dawley (SD), Zucker Diabetic fatty rats (ZDF) and rat insulin promoter-human islet amyloid polypeptide transgenic rats HIP). Rats (not treated with any drugs), n=36 per group at the start of the study, were analyzed after 4 months on a regular chow or high fat diet. SD rats gained more weight on the HFD, whereas HIP rats were slightly heavier on regular chow. 50% of the ZDF rats became unwell over time at 17-23 weeks of age. Plasma levels of amylase and lipase were higher at baseline in ZDF rats, and remained higher, relative to levels in the other rat strains. Plasma levels of MCP-1 and IL-6 varied substantially over time in all groups) and no correlation was noted between amylase, lipase, MCP-1 or IL-6 and histological analysis of findings in the exocrine pancreas (histology was reviewed by 3 separate pathologists). End of study pancreatic weights (normalized to body weight) were decreased in ZDF, SD-HFD and HIP rats, relative to other groups. Pancreatic exocrine inflammation (reduced size and number of acinar cells, necrosis, fibrosis, inflammatory cell infiltrate, more prominent ductal epithelial cells) was a common finding (up to 70% of all SD rats, 39% in HIP rats and 6% in ZDF rats) in all groups. Some SD rats also exhibited peri-islet inflammation. Abnormal ductal histology, including pancreatic duct glands (PDGs) was seen in 7/37 SD and ZDF rats, common enough to be considered a "normal histological feature" in these animals. No difference in histology was noted in when comparing head, body or tail of pancreas.
Roy and colleagues reported summary data for more than 70 preclinical toxicology studies examining the effects of saxagliptin and exenatide in rodents (up to 2 years) rabbits, dogs, and non-human primates (up to 12 months) at very high levels of clinical exposure. No histological evidence for drug-related pancreatitis or pancreatic neoplasms was noted in these studies The Glucagon-like Peptide-1-Based Therapeutics Exenatide and Saxagliptin Did Not Cause Detrimental Effects on the Pancreas in Mice, Rats, Dogs, and Monkeys Diabetes Obes Metab. 2014 Mar 26. doi: 10.1111/dom.12294
Koehler and colleagues have directly examined whether GLP-1 receptor activation modifies the proliferation or survival of human pancreatic cancer cells that express the endogenous GLP-1 receptor. The effect of Exenatide on human pancreatic cancer cell growth and survival was examined in studies using three different human pancreatic cancer cell lines that express the endogenous GLP-1receptor. Although Exenatide was able to activate several different signal transduction pathways in these cells, no significant effects of Exenatide were observed on stimulation of cell growth or cell survival in vitro. Furthermore, Exenatide had no effect on cell killing induced by multiple different cytotoxic agents in vitro. Tumor growth was also studied in vivo in mice treated with daily injections of Exenatide following implantation of human pancreatic tumors into nude mice. Exenatide did not stimulate cell proliferation or expansion of tumor mass in vivo. See Activation of Glucagon-Like Peptide-1 Receptor Signaling Does Not Modify the Growth or Apoptosis of Human Pancreatic Cancer Cells Diabetes 2006 55: 1369-1379.
Pancreatic Protein Synthesis In subsequent studies, Koehler explored the link between GLP-1R signaling and increased pancreatic mass in mice. The data show that while the mass of the pancreas increases by ~ 10% in mice treated with exenatide or liraglutide, DNA content or cell proliferation or edema do not change-rather, activation of GLP-1R signaling leads to increased protein synthesis in the pancreas. Furthermore, exendin-4 rapidly increased phosphorylation of proteins such as EIF2a, S6, and 4EBP1, all involved in control of protein synthesis. These effects of GLP-1R agonists required a functional GLP-1 receptor, were absent in Glp1r-/- mice and were eliminated by co-treatment of mice with rapamycin, an inhibitor of protein synthesis. Furthermore, although the relative abundance of the majority of proteins, including amylase and lipase, was not different after treatment with GLP-1R agonists, a number of proteins important for translation/protein syntesis were selectively increased after exendin-4 administration. See Glucagon-like peptide-1 receptor agonists increase pancreatic mass by induction of protein synthesis Diabetes published ahead of print October 2, 2014,doi:10.2337/db14-0883
Consistent with localization of Glp1r expression to the mouse and human exocrine pancreas, Hou et al demonstrated that Glp1r expression was identified in isolated pancreatic acini, and GLP-1 directly stimulated amylase release and cyclic AMP production, findings abolished in experiments using pancreatic tissue from Glp1r-/- mice. Loss of the Glp1r in Glp1r-/- mice did not perturb pancreatic morphology, or the basal expression/localization of amylase or phalloidin proteins in acinar cells; similarly basal mRNA transcripts levels for amylase, lipase, elastase, chymotrypsin, and ribonuclease was comparable in pancreatic lysates from Glp1r-/- vs. Glp1r+/+ mice. Native GLP-1 (30 pM-3 nM) stimulated amylase release from isolated acini from Glp1r-/- but not from Glp1r+/+ mice wheres VIP, A23187, CCK and cyclic AMP agonists (but not carbachol) produced comparable amylase secretion from Glp1r-/- and. Glp1r+/+ mice. No GLP-1-dependent potentiation of CCK-mediated amylase stiumulation was detected The Glucagon-Like Peptide-1 Receptor is Present in Pancreatic Acinar Cells and Regulates Amylase Secretion through cyclic AMP Am J Physiol Gastrointest Liver Physiol. 2015 Nov 5:ajpgi.00293.2015. doi: 10.1152/ajpgi.00293.2015
The putative relationship between GLP-1 receptor activation, exenatide, sitagliptin and the expression of genes and proteins that modules the risk of pancreatitis was examined in WT and Glp1r-/- mice. WT mice on a regular diet, a high fat diet, or following induction of diabetes with STZ, were treated, for both acute periods, and for several months with different GLP-1R agonists, metformin, or sitagliptin. Although GLP-1 receptor activation with exendin-4(exenatide) or liraglutide regulates gene and protein expression in the exocrine pancreas, GLP-1 receptor activation prior to or following the induction of experimental pancreatitis with the CCK analogue careulein had no effect on the development of experimental pancreatitis. Therapy with GLP-1R agonists actually induced a gene/protein expression program that would be predicted to be protective agaonst the development of pancreatitis. Moreover the molecular, biochemical and histological course and severity of pancreatitis was comparable in Glp1r-/- vs. Glp1r+/+ mice. See GLP-1 receptor activation modulates pancreatitis-associated gene expression but does not modify the susceptibility to experimental pancreatitis in mice Diabetes ; published ahead of print June 9, 2009, doi:10.2337/db09-0626
Forest and colleagues examined 300 ZDF diabetic rats treated with placebo, metformin, or 3 doses of sitagliptin for up to 14 weeks. No histological evidence for pancreatic inflammation, proliferation in the ductual inflammation, or neoplasia, was noted, following analysis of more than 5,000 sections of the pancreas Characterization of the Exocrine Pancreas in the Male Zucker Diabetic Fatty Rat Model of Type 2 Diabetes Mellitus Following 3 Months of Treatment with Sitagliptin Endocrinology en.2013-1781
Nyborg and colleagues examined the chronic effects of liraglutide in 3 species, rats, mice and monkeys for up to 2 years, exposed to a range of liraglutide doses. Histological abnormalities were quantified in liraglutide- vs. placebo treated groups encompassing hundreds of animals. Monkeys were doses for up to 87 weeks with no evidence for preneoplastic lesions, pancreatitis, or pancreatic cancer despite exposure to plasma liraglutide concentrations up to 60 fold higher than those obtained in clinical studies in humans. Rats were treated for up to 2 years. No significant inflammatory or preneoplastic lesions were detected in the pancreata of mice, rats, and monkeys. The Human GLP-1 Analog Liraglutide and the Pancreas: Evidence for the Absence of Structural Pancreatic Changes in Three Species Diabetes. 2012 May;61(5):1243-9.
Ellenbroek treated normal diet- or high fat fed mice with liraglutide for 1 or 6 weeks. Increased acinar cell proliferation and size was detected in mice on a normal chow diet, but not in mice on a high fat diet after 6 weeks of twice daily liraglutide. Glucagon-like peptide-1 receptor agonist treatment reduces beta cell mass in normoglycaemic mice Diabetologia. 2013 Jun 16
Tatarkiewicz and colleagues examined the effects of exenatide in normal and diabetic mice treated with continuous exenatide infusions for 4 weeks and in mice and rats treated with acute exenatide. Experimental pancreatitis was induced using caerulein or sodium taurocholate and plasma markers of inflammation and pancreatic histology was examined. Acute or chronic exenatide administration had no effect on plasma amylase or lipase in the absence of inflammation, and in some experiments, plasma amylase and lipase actually decreased after exenatide treatment, in the presence or absence of pancreatitis. Exenatide had no consistent effect on inflammatory mediators, and at the highest doses employed, actually decreased histological markers of inflammation in both experimental models. The rates of ductal proliferation were also examined and were unchanged after exenatide treatment in ob/ob mice however exenatide increased the expression of the protective gene, PAP. See Exenatide does not evoke pancreatitis and attenuates chemically-induced pancreatitis in normal and diabetic rodents Am J Physiol Endocrinol Metab. 2010 Oct 5
A subsequent study examined the effects of treating Zucker diabetic fatty rats with a range ( 6, 40 and 250 µg/kg/day ) of exendin-4 doses, twice daily for 13 weeks. No significant effects of exendin-4 treatment were noted in assessments of lipase concentrations, pancreatic weight, pancreatic histology, ductal cell proliferation or apoptosis. No evidence of drug-induced pancreatitis in rats treated with exenatide for 13 weeks Diabetes Obes Metab. 2012 Nov 19. doi: 10.1111/dom.12040
Analysis of the biological activity of exendin-4 in guinea pig pancreatic exocrine slices revealed that exendin-4 stimulates cyclic AMP release through incompletely characterized mechanisms as outlined in Isolation and characterization of exendin-4, an exendin-3 analogue, from Heloderma suspectum venom. Further evidence for an exendin receptor on dispersed acini from guinea pig pancreas. J Biol Chem. 1992 Apr 15;267(11):7402-5. Subsequent experiments revealed that although exendin-4 alone did not stimulate amylase release, exendin-4 potentiated amylase release induced by CCK, carbamylcholine, bombesin or a calcium ionophore, A23187 Exendin-4, a new peptide from Heloderma suspectum venom, potentiates cholecystokinin-induced amylase release from rat pancreatic acini. Regul Pept. 1992 Sep 22;41(2):149-56. These interactions of exendin-4 with pancreatic acinar cells are also seen with native GLP-1, which increased cyclic AMP release in comparable studies, actions which were blocked by the GLP-1R antagonist exendin(9-39); similarly, binding sites for both exendin-4 and GLP-1 were detected in experiments using radiolabelled peptides and guinea pig pancreatic acinar cells Truncated glucagon-like peptide-1 interacts with exendin receptors on dispersed acini from guinea pig pancreas. Identification of a mammalian analogue of the reptilian peptide exendin-4. J Biol Chem. 1992 Oct 25;267(30):21432-7. and Use of 125I-[Y39]exendin-4 to characterize exendin receptors on dispersed pancreatic acini and gastric chief cells from guinea pig. Regul Pept. 1994 Aug 31;53(1):47-59
There is very little information available about the possible effects of GLP-1/exendin-4 on the gall bladder or biliary tract. Preclinical studies in rats demonstrate that GLP-1/exendin-4 stimulates cholangiocyte growth, and that proliferating cholangiocytes may be capable of actually synthesizing GLP-1 Glucagon-like peptide-1 and its receptor agonist exendin-4 modulate cholangiocyte adaptive response to cholestasis. Gastroenterology. 2007 Jul;133(1):244-55. Subsequent studies demonstrated that exendin-4 is also capable of exerting anti-apoptotic effects on rat cholangiocytes cultured in vitro, and in a bile duct ligation/toxin (Ccl4)-induced model of cholangiocyte apoptosis in vivo Exendin-4, a Glucagon-Like Peptide 1 receptor agonist, protects cholangiocytes from apoptosis. Gut. 2008 Oct 1. [Epub ahead of print]. Cholangiocytes from rats with bile duct ligation express Pdx-1 and GLP-1R activation enhances nuclear Pdx1 protein expression in rat cholangiocytes. IGF-1 expression was also induced following exendin-4 treatment of rat cholangiocytes and the ability of exendin-4 to induce IGF-1 and VEGF expression and cholangiocyte proliferation was impaired following Pdx1 knockdown Pancreatic Duodenal Homeobox-1 de novo expression drives cholangiocyte neuroendocrine-like transdifferentiation J Hepatol. 2010 Oct;53(4):663-70. As noted below in the section on cancer, GLP-1R activation reduced the growth and enhanced the apoptosis of human cholangiocarcinoma cell lines both in vitro and in mice in vivo Effect of the GLP-1 Analog Exendin-4 and Oxaliplatin on Intrahepatic Cholangiocarcinoma Cell Line and Mouse Model. Int J Mol Sci. 2013 Dec 13;14(12):24293-304.
The putative mechanism through which GLP-1R agonists may act on the exocrine pancreas remains uncertain, and pancreatitis has not been commonly described in hundreds of preclinical studies of GLP-1R agonists or DPP-4 inhibitors. Infusion of GLP-1 inhibits pancreatic exocrine secretions in short term studies of normal human subjects Truncated GLP-1 (proglucagon 78-107-amide) inhibits gastric and pancreatic functions in man. Dig Dis Sci. 1993 Apr;38(4):665-73. These effects are likely indirect, due to the effects of GLP-1 on reduction of gastric emptying, and hence reduced transit of food into the small bowel.
Treatment of Sprague Dawley rats with exendin-4 once daily for 75 days produced no changes in amylase, but a 2-fold increase in serum lipase was noted, in associated with histological changes consistent with mild pancreatitis in some acini. Remarkably, the exendin-4-treated rats exhibit a profound reduction in body weight gain, with the control rats starting at ~250 grams and ending up at 600 grams after 9 weeks, whereas rats treated with exendin-4 started at ~240 grams and ended up at ~ 390 grams. No other anti-diabetic agents were included as controls, and no pair-fed controls were studied to assess the effects of marked weight loss alone, a know risk factor for pancreatitis, in the study. The authors stated that "the histological differences observed between controls and exendin-4-treated animals were not very striking". Some exendin-4-treated rats exhibited morphological changes consistent with acinar fibrosis, and focal acinar degeneration and inflammation, as outlined in Biochemical and histological effects of exendin-4 (exenatide) on the rat pancreas Diabetologia. 2009 Sep 13. [Epub ahead of print]. In an accompanying Editorial, Butler and colleagues summarized the available evidence, and based on their interpretation of the available animal data, recommended that therapy with GLP-1 mimetics only be used in conjunction with metformin Glucagon-like peptide-1 therapy and the exocrine pancreas: innocent bystander or friendly fire? Diabetologia. 2009 Nov 6. [Epub ahead of print]. Transgenic over-expression of exendin-4 has also been associated with the development of an immune response against the preproexendin-4 transgene but not exendin-4 alone in mice, in association with tissue infiltrates involving multiple organs, including the, heart, liver, kidney and pancreas Lymphocytic infiltration and immune activation in metallothionein promoter-exendin-4 (MT-Exendin) transgenic mice Diabetes. 2006 Jun; 55 (6): 1562-70
Matveyenko and colleagues demonstrated adverse effects of sitagliptin on pancreatic histology in HIP-transgenic rats with overexpression of human amylin in the beta cells of transgenic animals. Groups of rats were treated with either sitagliptin alone, metformin, or both drugs together. Significantly increased ductal turnover was observed in non-treated transgenic rats, and sitagliptin increased whereas metformin decreased ductal turnover. Sitagliptin was also associated with increased ductal metaplasia, and one sitagliptin-treated rat developed focal pancreatitis. See Beneficial endocrine but adverse exocrine effects of sitagliptin in the human islet amyloid polypeptide transgenic rat model of type 2 diabetes: interactions with metformin Diabetes. 2009 Jul;58(7):1604-15.
In a subsequent study Pancreatic duct replication is increased with obesity and type 2 diabetes in humans Diabetologia. 2009 Oct 21. [Epub ahead of print], the same authors studied rates of ductal replication in pancreata from autopsy studies of 45 humans subdivided into 4 groups; lean, obese, non-diabetic, and subjects with type 2 diabetes. Obesity (BMI greater than 27) was associated with a 10-fold increase in the rate of ductal replication as assessed by staining with Ki67. Cytokeratin was used as a marker for ductal cells. Lean subjects with type 2 diabetes exhibited a 4-fold increase in ductal cell replication compared to lean non-diabetic controls. Obese individuals with diabetes did not exhibit a further increase in ductal cell replication. The autopsy series represents a subgroup selected from a previously published analysis of beta cell mass and replication Beta-cell deficit and increased beta-cell apoptosis in humans with type 2 diabetes Diabetes. 2003 Jan;52(1):102-10. The authors suggest a note of caution in regard to therapeutic strategies based on incretin action that are used to treat human diabetics as these agents (GLP-1R agonists and DPP-4 inhibitors) have been shown to increase ductal replication in rodents. Intriguingly, a similar analysis of pancreatic ductal turnover in human subjects subjected to one or 2 pancreatic surgeries, who exhibited progressive increases in blood glucose to the frankly diabetic range, did not reveal any increases in pancreatic ductal replication Partial pancreatectomy in adult humans does not provoke beta-cell regeneration Diabetes. 2008 Jan;57(1):142-9. Although the Butler paper Pancreatic duct replication is increased with obesity and type 2 diabetes in humans Diabetologia. 2009 Oct 21. [Epub ahead of print] and Editorial Glucagon-like peptide-1 therapy and the exocrine pancreas: innocent bystander or friendly fire? Diabetologia. 2009 Nov 6. [Epub ahead of print] speculates that GLP-1 may induce chronic pancreatitis leading to enhanced ductal cell replication and an increased risk of pancreatitis, careful quantitative analysis of pancreatic tissue from 43 human subjects with chronic pancreatitis did not reveal any differences in ductal replication between subjects with pancreatitis vs. control subjects without pancreatitis. These authors speculate that the high rates of ductal turnover seen in many rodent studies may not be representative of ductal replication rates in humans. See Figure 6 in Reduced pancreatic volume and beta-cell area in patients with chronic pancreatitis Gastroenterology. 2009 Feb;136(2):513-22.
Gier and colleagues examined the effects of exendin-4 on human pancreatic ductal cell replication and on formation and number of dysplastic foci in mice with Kras-induced pancreatic dysplasia. WT rats treated with once daily injections of exendin-4 exhibited a 'relative' increase in pancreatic weight, no pancreatitis, but an expansion of the PDG (pancreatic duct gland compartment), with a 4-fold increase in ductal cell replication. Treatment of the Pdx-1Kras for 12 weeks with exendin-4 significantly increased pancreatic weight, increased the histological evidence for pancreatitis, increased ductal cell proliferation, increased plasma lipase activity, and increased inflammation, fibrosis, and metaplastic changes. Immunohistochemistry demonstrated GLP-1R-positive ductal cells but no GLP-1R expression was detected in pancreatic acinar cells. GLP-1R expression was also detected in PanIN lesions (pancreatic intraepithelial lesions) and exendin-4 increased cyclinD1, ERK1/2 and proliferation of human ductal cells. Concomitant treatment with metformin reduced the effects of exendin-4 on ductal proliferation. Chronic GLP-1 Receptor Activation by Exendin-4 Induces Expansion of Pancreatic Duct Glands in Rats and Accelerates Formation of Dysplastic Lesions and Chronic Pancreatitis in the KrasG12D Mouse Model Diabetes published ahead of print January 20, 2012, doi:10.2337/db11-1109
The incidence of pancreatic cancer is known to be increased in diabetic subjects, and in some studies, the more advanced duration of diabetes (requiring insulin therapy) together with gallstones/gall bladder disease further increased the relative risk in retrospective association studies Gallstones, a cholecystectomy, chronic pancreatitis, and the risk of subsequent pancreatic cancer in diabetic patients: a population-based cohort study J Gastroenterol. 2012 Oct 3. [Epub ahead of print]
On Sept 25 2009, the FDA posted the following notice about case reports of pancreatitis in patients treated with sitagliptin.
Sitagliptin (marketed as Januvia and Janumet) - acute pancreatitis
FDA notified healthcare professionals and patients of revisions to the prescribing information for Januvia (sitagliptin) and Janumet (sitagliptin/metformin) to include information on reported cases of acute pancreatitis in patients using these products. Eighty-eight post-marketing cases of acute pancreatitis, including two cases of hemorrhagic or necrotizing pancreatitis in patients using sitagliptin, were reported to the Agency between October 2006 and February 2009. It is recommended that healthcare professionals monitor patients carefully for the development of pancreatitis after initiation or dose increases of sitagliptin or sitagliptin/metformin. Sitagliptin has not been studied in patients with a history of pancreatitis. Therefore, it is not known whether these patients are at an increased risk for developing pancreatitis and the medication should be used with caution and with appropriate monitoring in patients with a history of pancreatitis. Considerations for healthcare professionals, information for patients, and a Data Summary are provided.
GLP-1 and cancer
Ligumsky and colleagues examined the effects of GLP-1 and exendin-4 on breast cancer growth and survival. Although the classical GLP-1 receptor was not detected in breast cancer cell lines, the GLP-1R agonists reduced breast cancer cell (MCF-7, MDA-MB231 and MDA-MB-468) growth and increased apoptosis, both in cells cultured in vitro, and in breast cancer tumors propagated in athymic nude mice treated with daily exendin-4 (5 days a week for 6 weeks) or with continuous infusion pumps for 4 weeks in vivo. GLP-1R agonists increased accumulation of cAMP, and cAMP inhibition reversed the effects of GLP-1/exendin-4 on breast cancer cell growth. See The peptide-hormone glucagon-like peptide-1 activates cAMP and inhibits growth of breast cancer cells. Breast Cancer Res Treat. 2011 Jun 3.
Koehler and Kain examined the effects of exendin-4 on colon cancer cell growth in cells propagated in vitro, and in syngeneic tumors implanted in mice in vivo. Mouse CT26 colon cancer cells expressed a full length functional GLP-1R coupled to cAMP activation. Both forskolin and exendin-4 reduced cell growth and increased apoptosis in CT26 cells. Exendin-4 reduced colony formation in soft agar and enhanced apoptosis in tumor cells in mice. The ability of exendin-4 to enhance colon cancer cell death was enhanced in the presence of cytotoxic agents. GLP-1R activation inhibited Erk1/2 activation and inactivated GSK3, athough different mechanisms. See Glucagon-Like Peptide-1 Receptor Activation Inhibits Growth and Augments Apoptosis in Murine CT26 Colon Cancer Cells Endocrinology. 2011 Jul 19.
Chen and colleagues studied the effects of exendin-4, with and without oxaliplatin, on the growth and apoptosis of human cholangiocarcinoma cells (HuCCT1) in vitro and in mice in vivo. GLP-1/exendin-4 reduced colony formation and cell proliferation in vitro and exendin-4 treatment once daily for 1 week followed, in some instances, by weekly oxaliplatin administration) of Balb/C mice with subcutaneous HuCCT1 tumors GLP-1R activation (exendin-4 alone) reduced the growth rate of tumors in vivo, and further reduced tumor growth in mice receiving chemotherapy. These findings were associated with enhanced apoptosis and reduced cell migration of human cholangiocarcinoma cell lines both in vitro and in mice in vivo Effect of the GLP-1 Analog Exendin-4 and Oxaliplatin on Intrahepatic Cholangiocarcinoma Cell Line and Mouse Model. Int J Mol Sci. 2013 Dec 13;14(12):24293-304.
Thyroid C cells and calcitonin
Preclinical studies demonstrate that rodent, specifically mouse and rat thyroid calcitonin-producing C cells express a functional GLP-1 receptor. Indeed several studies employing rodent C cell lines demonstrate that GLP-1R activation in vitro leads to rapid stimulation of calcitonin secretion, actions that are attenuated by blocking the GLP-1 receptor with the antagonist exendin(9-39) Effects of glucagon and glucagon-like peptide-1-(7-36) amide on C cells from rat thyroid and medullary thyroid carcinoma CA-77 cell line Endocrinology. 1996 Sep;137(9):3674-80 Effects of glucagon and glucagon-like peptide-1-(7-36) amide on C cells from rat thyroid and medullary thyroid carcinoma CA-77 cell line and Expression of glucagon-like peptide 1 receptor in a murine C cell line: regulation of calcitonin gene by glucagon-like peptide 1 FEBS Lett. 1996 Sep 16;393(2-3):248-52. In contrast, functional GLP-1 receptors coupled to calcitonin secretion have not been detected in human C cell lines.
The role and importance of GLP-1 receptor activity in rodent C cells is likely related to the gut-bone axis and control of bone resorption. In rodents, and to some extent in humans many gastrointestinal peptides inhibit bone resorption in the postprandial state Potential role of pancreatic and enteric hormones in regulating bone turnover. J Bone Miner Res. 2005 Sep;20(9):1497-506. Indeed administration of the GLP-1R agonist exendin-4 increases calcitonin gene expression in WT mice, whereas Glp1r-/- mice exhibit increased bone resorption and reduced calcitonin expression in the thyroid The murine glucagon-like peptide-1 receptor is essential for control of bone resorption Endocrinology. 2008 Feb;149(2):574-9. Nevertheless, in contrast to the biology in rodents, GIP or GLP-1 do not inhibit bone resorption in human subjects whereas the related peptide GLP-2 is a potent inhibitor of bone resorption Role of gastrointestinal hormones in postprandial reduction of bone resorption J Bone Miner Res. 2003 Dec;18(12):2180-9.
Continuous activation of the GLP-1 receptor in rats and to a lesser extent in mice produces C-cell hyperplasia, periodically associated, after sustained administration, with the subsequent development of medullary thyroid cancer. Moreover, acute administration of any GLP-1R agonist produces a rapid increase in plasma calcitonin levels in rodents. In contrast, studies with monkeys demonstrate a) no increase in plasma calcitonin levels and b) no C-cell hyperplasia and c) no MTC despite continuous prolonged GLP-1R agonist administration. Similarly, administration of liraglutide once daily to thousands of human subjects with diabetes or obesity did not result in significant increases in plasma calcitonin over time. Taken together, the available data demonstrate a major species-specific difference in the biology of GLP-1 receptor activity in the rodent vs primate thyroid gland. See Glucagon-like peptide-1 receptor agonists activate rodent thyroid C-cells causing calcitonin release and C-cell proliferation Endocrinology 2010 Apr;151(4):1473-86. C-cell hyperplasia was not observed following continuous exenatide or intermittent liraglutide dosing for 13 weeks in Glp1r-/- mice, whereas control mice exhibited calcitonin increases and C-cell hyperplasia with exenatide and liraglutide. Evaluation of RET activation using a phospho-RET antibody and immunohistochemistry revealed no evidence for RET activation in the thyroid glands of WT mice treated with liraglutide, however increased levels of the mTOR-dependent S6 protein were detected in the thyroid glands from the same experiments. GLP-1 Receptor Agonists and the Thyroid: C-Cell Effects in Mice Are Mediated via the GLP-1 Receptor and not Associated with RET Activation Endocrinology January 10, 2012 en.2011-1864.
Qualitatively similar results have been detected in preclinical studies with the long acting GLP-1R agonist dulaglutide, which had no effect on C-cell proliferation, C-cell volume, or serum levels of calcitonin when dosed at relatively high levels ( 8.15 mg/kg twice weekly in male monkeys for 52 weeks which achieved plasma drug levels ~500-fold greater than maximal human exposure). Dulaglutide was almost certainly active as the monkeys exhibited a sustained reduction in body weight Effects of Dulaglutide on Thyroid C-Cells and Serum Calcitonin in Male Monkeys Endocrinology. 2015 Apr 10:en20141717. In contrast, rats were much more susceptible to C-cell proliferation, exhibiting hyperplasia and medullary thyroid carcinoma at lower multiples of drug exposure, whereas WT and RASH2 transgenic mice exhibited minimal C-cell changes after 26 weeks of dulaglutide.Chronic Toxicity and Carcinogenicity Studies of the Long-Acting GLP-1 Receptor Agonist Dulaglutide in Rodents Endocrinology. 2015 Apr 10:en20141722
Nevertheless, the human GLP-1 receptor has been shown, using immunocytochemical techniques to be expressed in subsets of thyroid tumor cells from patients with C cell hyperplasia, medullary thyroid cancer, and a smaller proportion of papillary thyroid cancers. The functional significance of these findings, if any, has not yet been ascertained, and the sensitivity and specificity of the antisera used to detect the GLP-1R in these studies remains uncertain. Glucagon Like Peptide-1 Receptor Expression in the Human Thyroid Gland. J Clin Endocrinol Metab. 2012 Jan;97(1):121-31
Boess and colleagues examined GLP-1R expression and action in rodent and human thyroid glands and in primary thyroid cell cultures. Rat cultures were responsive to TSH, and calcium promoted calcitonin secretion; human thyroid cultures were much less responsive and more variable. Glp1r mRNA transcripts were not consistently detected in RNA from rat or human thyroids or from primary thyroid cultures. In situ hybridization co-localized Glp1r and calcitonin RNA in rat thyroid C cells. Exendin-Cy5 detected GLP-1R binding on the cell surface of transfected cells, but no positive thyroid cells were visualized in binding studies using this probe. Liraglutide and taspoglutide increased calcitonin release in rat thyroid cultures but not in studies with human thyroid cultures. Effect of GLP-1R agonists taspoglutide and liraglutide on primary thyroid C-cells. J Mol Endocrinol. 2013 Mar 5
Pach and colleagues reported the use of an exendin-4-based imaging ligand (Lys40(Ahx-HYNIC-99mTc/EDDA)NH2]-Exendin-4) for visualization of recurrent or disseminated medullary thyroid cancer in 4 human subjects with elevated calcitonin levels (two with MEN, two with sporadic MTC). The extent of GLP-1 receptor expresion (mRNA or protein) in human tumor tissue excised from these patients was not evaluated. No control patients without MTC were studied. Glucagon-Like Peptide-1 Receptor Imaging with [Lys (40) (Ahx-HYNIC- (99 m) Tc/EDDA)NH 2 ]-Exendin-4 for the Diagnosis of Recurrence or Dissemination of Medullary Thyroid Cancer: A Preliminary Report Int J Endocrinol. 2013;2013:384508
Waser and colleagues used in situ radiography and iodinated peptides to study GLP-1 and GIP receptor binding capacity in rodent and human thyroid glands. GLP-1 but not GIP receptor binding activity was easily detected in normal rodent thyroid glands, and in rat medullary thyroid cancer cells; GIP receptors were also detected in rat medullary thyroid cancer but not in specimens of C cell hyperplasia. GIP and GLP-1 receptor binding was not detected in normal human thyroid glands. 27% of human medullary thyroid cancers contained GLP-1 receptor binding sites, whereas GIP receptor binding sites were more numerous and higher density, with 89% of medullary thyroid cancers exhibiting GIP receptor positivity. The authors also detected GIP receptors in human TT thyroid cells, that do not express the GLP-1R. Incretin Receptors in Non-Neoplastic and Neoplastic Thyroid C Cells in Rodents and Humans: Relevance for Incretin-Based Diabetes Therapy Neuroendocrinology. 2011 Sep 2.
Additional longitudinal data from serial measurements of calcitonon levels in over 5,000 individuals with either diabetes or obesity treated with liraglutide was presented by Hgedus et al. Non-stimulated calcitonin measurements were measured every 3 months, in liraglutide vs. control subjects for up to 2 years in 8 phase 3 clinical trials for diabetes, and 1 phase 2 trial for obesity. No clinically significant shift in mean calcitonin levels was observed across the entire study population and no differences were seen in calcitonin levels in subjects treated with exenatide vs. liraglutide. Several cases (6) of C-cell hyperplasia were identified, most with elevated baseline calcitonin levels, and one case of MTC was described in a non-liraglutide-treated patient. Liraglutide did not produce further increases in calcitonin levels even in subjects with baseline CT elevations. See GLP-1 and Calcitonin Concentration in Humans: Lack of Evidence of Calcitonin Release from Sequential Screening in over 5000 Subjects with Type 2 Diabetes or Nondiabetic Obese Subjects Treated with the Human GLP-1 Analog, Liraglutide. J Clin Endocrinol Metab. 2011 Jan 5. [Epub ahead of print]
